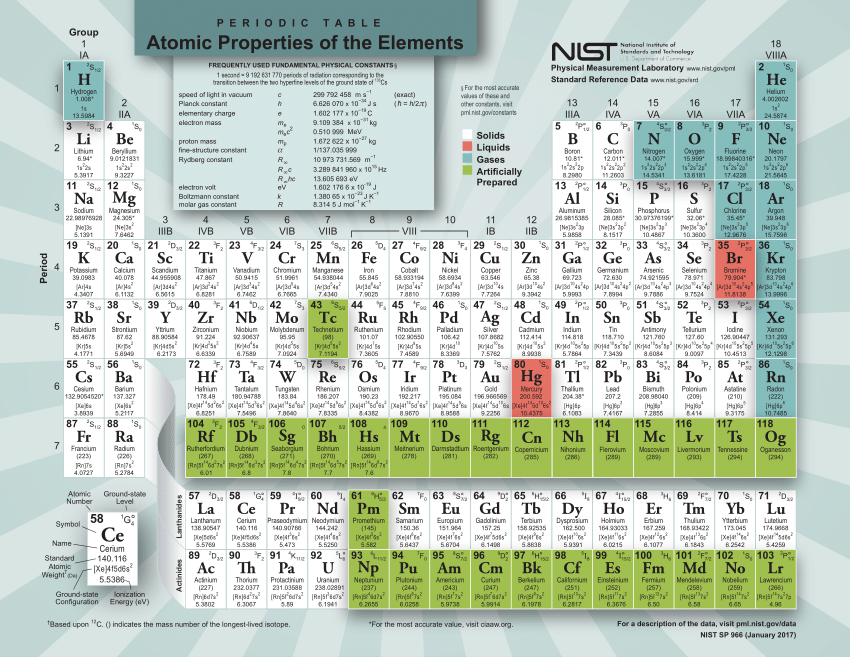
There are x 1021 atoms of chlorine in 0.501 moles of TiCl4. Round the answer to the correct number of significant figures and enter the number only. 1 pts D Question 6 Calculate the number of atoms. It is obtained chiefly from the mineral cassiterite, which contains tin dioxide. Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. How many moles of BaCl₂ was present at the start of the reaction? Enter the number only, with no units and with the correct number of significant figures. Tin is a post-transition metal in group 14 of the periodic table. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. Question 5 1 pts Silver nitrate reacts with barium chloride in the following precipitation reaction (balanced): 2 AgNO3 + BaCl₂ → 2 AgCl + Ba(NO3)₂ Gravimetric analysis of the products showed that there were 0.7050 moles of silver chloride and 0.3525 moles of barium nitrate. Ochlorine tribromide Olithium monoxide lithium disulfide Diron(III) sulfate cesium sulfide lithium sulfide AN The percent composition of titanium in TiCl, is % Question 4 1 pts Which of the following names do not follow the rules for nomenclature? Choose all the incorrectly named compounds in the list. Our final answer is expressed to four significant figures. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum.

Round the answer to four significant figures and enter the number only. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Au: Molar Mass (g/mol) Au (Gold) 1 × 196.966569 196.966569. 3.987 molAl × 26.98gAl 1 molAl 107.6gAl 3.987 mol Al × 26.98 g Al 1 mol Al 107.6 g Al.
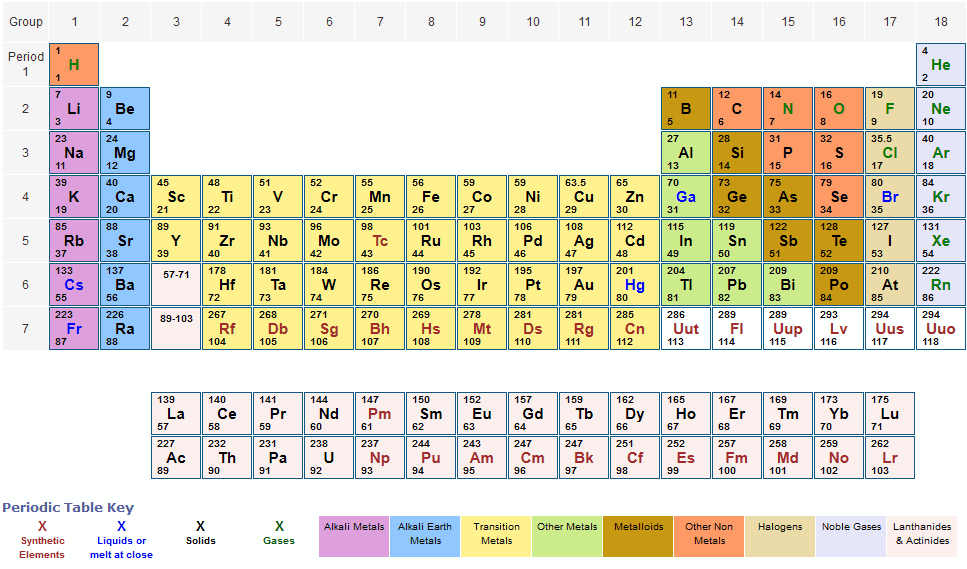
Sy10028402/take Question 3 1 pts Use the periodic table above to calculate the percent composition. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. D Question 2 2 pts Use the above periodic table to calculate the mass in grams of S2 needed to react with 4.90 g of C8H18 in the balanced equation: 2 C8H18 + 25 S₂→→ 16 CS₂ +18 H₂S Enter the numerical value only, with no units and with the correct number of significant figures. The molar mass of beryllium sulfide is amu. (289) (288)) Question 1 1 pts Use the periodic table above to calculate the molar mass. Atomic Mass in the Periodic Table of Elements.
